Update from the Field: Keep Calm and Test On
Posted by Dave Dyson, Agronomist on November 23, 2021

Soil sampling can be a long and laborious process, but it is necessary to keep calm and perform soil tests to evaluate fertility levels. Think of soil testing as your fuel gauge on a long road trip. On a road trip, we endure long window times, smelly aromas from the back seat, and routinely check our fuel level. Similarly, soil testing can be a long and boring process, but along the way we accurately gauge our fertility levels so we can properly replenish nutrients and successfully get from point A to point B.
It is important to measure the fertility levels of our soil to be able to make informed decisions for the application of fertilizer. Fertility levels can build up in the soil over time with repeated applications of fertilizer and performing a soil test can ensure you are not applying too much fertilizer. Conversely, micronutrient levels are notoriously low in the Midwest, Figure 1, but can drastically increase yield when brought to proper levels.

Figure 1: Zinc can be a major contributor to yields. According to A&L Great Lakes Laboratories the amount of zinc in all the soil tests they process is on a downward trend. The amount of zinc needed to be applied to a cropping system is very low, so knowing the exact amount in your soil is crucial for a correct recommendation.
Soil pH is considered a master variable in the soil as it affects many chemical processes. It specifically impacts the availability of nutrients by controlling the chemical forms of the nutrients and influencing the chemical reactions they undergo. The nutrients needed to grow our crops are most available in the soil between the 6-7 pH range, Figure 2. Correcting the soil pH is like steering a ship small course changes are much easier than drastic adjustments.
Soil pH is defined as a measure of acidity and alkalinity in the soil. Soil pH levels range from 0 to 14, with 7 being neutral. We measure the activity of hydrogen ions in a soil/water solution and usually falls within a range of 3-10 pH. Acid soils have a pH below 7 and alkaline soils have a pH above 7. This is a negative logarithmic scale, so as you increase from 5 to 6 pH, you have a drop in the amount of hydrogen ions by a factor of 10. As a result, changing the pH a little is good but too much can cause serious damage to your field. Therefore, soil tests are critical before amending the soil’s ;pH.
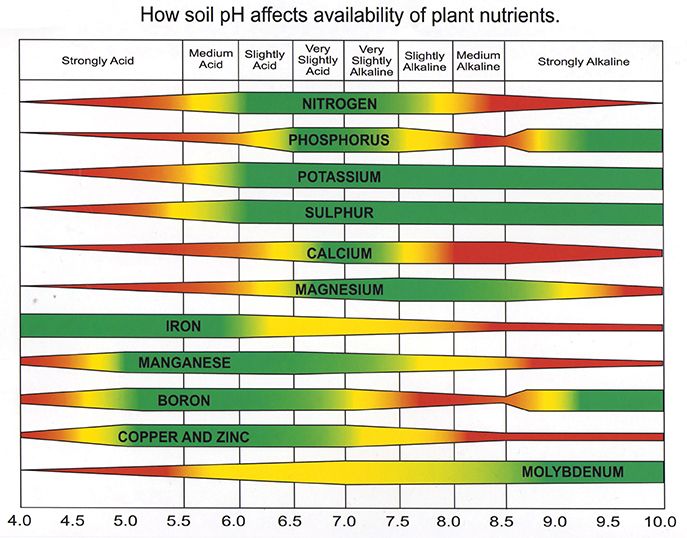
Figure 2: Nutrient availability vs. pH chart by Purdue University
Albert Einstein said it best, “If you want to know the future, look at the past.” If we want to move forward to receive a positive return on investment with our cropping system, we must look back at where our soil fertility levels have been. The fertility levels of our soil can tell us how much and what type of fertilizer to apply in the future. If you have questions about performing or interpreting a soil test, contact your Trusted Ag Advisor from The Andersons.
FOR MORE INFORMATION:
Please complete the form, and we’ll get you in touch with your Territory Manager from The Andersons.

Dave Dyson is a regional agronomist for The Andersons’ Farm Centers which are located throughout Ohio, Michigan, and Indiana. He is an Indiana native and grew up on a dairy farm in Miami County. A graduate of Purdue University with a degree in Crop & Soil Science, Dave has a deep knowledge of various agronomic topics and is committed to helping growers improve their crops. If you have any questions, Dave can be reached at david_dyson@andersonsinc.com.
© 2021 The Andersons, Inc. All Rights Reserved.


